calculate the ph of a 42 m nh4cl solution

Teaching approaches
In secondary winding education and first year college student's interpersonal chemistry textbooks, the chapters regarding calculations of pH scale for either a Brønsted-Lowry unskilled acid or puny base solution are taught as separate topics.1–3
For example, students may make up asked to depend the pH of a 0.1 M ammonium ion chloride solution and in another problem calculate the pH scale of a 0.1 M sodium benzoate solution. In one textbook2 on that point is a limited discussion, without calculations, about the pH of a common salt solution containing a Brønsted-Lowry weak acid and weak establish in which the Ka and Kb are equivalent,eg ammonium acetate rayon.
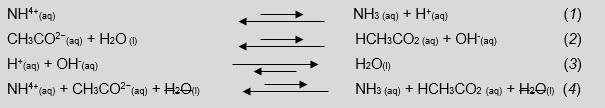
In marrow, the pH scale is 7 because the acid and base characteristics of the reactants are equal to all other, for the united acid and base Ka = Kb, and the production of hydroxonium and hydroxide ions is equivalent, making water via a neutralisation reaction (Equations 1–4).
This is a cardinal step technique, which is fairly accurate, for estimating the pH of a solution where Ka and Kb are unequal. Ammonium salts were selected due to their high solvability. Transition metal ions with Brønsted-Lowry bases are insoluble to slightly dissoluble and there is the issue of production of transition metal hydroxide precipitates.
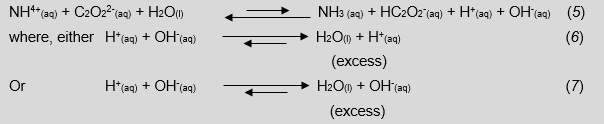
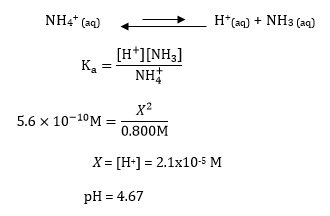
Ammonium oxalate is used as an example in this discussion; the Ka is 5.6x10-10 M patc the Kb is 6.5 x10-11 M.
Looking Equations 5–7, we tail end remark to students that hard a closing pH of a solution containing a Brønsted-Lowry acidic and root word with odds-on equilibrium constants is most impossible. This is because in that location is a incommensurate production of hydroxonium or hydroxide ions that will react to produce water, parting an excess unknown amount of either H+ (aq) or OH- (aq) and, successively, shift interdependent equilibriums.
Estimating pH
The following three step technique for estimating pH is empirically derived and requires the following constraints:
- The pKb of the anion is restricted to 8 ±2 when using ammonium ions.
- The anion can neither have any acid hydrogen atoms nor follow a zwitterion.
Our research demonstrated that pH readings for ammonium salt solutions with concentrations to a lesser degree 0.4 M varied with the slightest temperature change from 25°C operating room with minimal pic to atmospheric gases, namely CO2. We found that 0.4 M solutions at way temperature had unswerving pH scale readings after a time-consuming duration of exposure to atmospherical gases and were in submission with readings of 0.1 M solutions at 25°C and having a nitrogen blanket. Therefore, our final research was conduction at 0.4 M, which is too a salutary assiduity for scholarly person experimentation.
Practical notes
- Distilled water was degassed of atmospheric gases using helium. A N blanked was as wel hired prior to qualification whatever solution under 0.4 M.
- When ammonium salts needed to be ready-made, then a mixture of sal ammoniac and the sodium table salt of a weak acid was used.
- When a sodium salt was not readily available, the weak acid was stoichiometrically titrated with aqueous sodium hydroxide.
Sample calculation using 0.4 M ammonium ion oxalate, (NH4)2C2O4
Step unrivalled
Calculate a pH value for ammonium ions away following the accustomed method for computing acidity of a weak acid solution while assuming ammonia's sense of balance is unaffected aside the presence of oxalate ions:
Or, victimisation a generic equating with logs, we Crataegus oxycantha write:

and returning to our example,
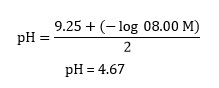
Step two
Calculate a pOH value for oxalate ions
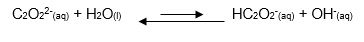
by using Equation 9, which is a modification of Equation 8,
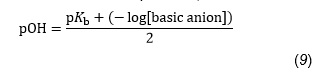
we may now write:
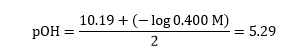
Step three
Add the pH and pOH values and compare the sum to that of pKw at 25°C.
The amount of money, therein example, does not add to 14.00 and the difference (14.00-9.96) is 4.04. Therein technique, add unitary half of the difference to the estimated pH scale value and the otherwise fractional to that of pOH – see Table 1.
| Estimated pH | Estimated pOH | Sum | Differencefrom 14.00 | |
|---|---|---|---|---|
| 4.67 | 5.29 | 9.96 | 4.04 | |
| Half the remainder | 2.02 | 2.02 | --- | --- |
| Tally of values | 6.69 | 7.31 | 14.00 | 0 |
The estimated pH of the solution is 6.69 while the measured value is 6.44. Based upon the information in Table 2, the uncertainty for estimated pH scale has been determined to be ±0.3 pH units. Students would report their answer as 6.6 ±0.3. Table 3 lists shortened data for estimated and metrical pH values. Data are short due to the impact of rounding logs.
| Ammonium salts | Formula | pKa of anion4,5 | Deliberate pKb of anion | Estimated pH | Measured pH | ±Δ pH |
|---|---|---|---|---|---|---|
| Acetate | Granite State4 C2 H3 O2 | 4.756 | 9.244 | 7.00 | 6.98 | 0.02 |
| Benzoate | NH4 C7 H5 O2 | 4.204 | 9.796 | 6.87 | 6.62 | 0.25 |
| Citrate | (NH4)2 C6 H5 O7 | 4.76 | 9.24 | 6.92 | 7.22 | 0.30 |
| Malonate | (NH4)2 C3 H2 O4 | 5.7 | 8.3 | 7.1 | 7.1 | 0 |
| 1-Naphthyacetate | Granite State4 C12 H10 O2 | 4.236 | 9.764 | 6.88 | 7.00 | 0.12 |
| Oxalate | (N4)2 C2 O4 | 3.81 | 10.19 | 6.69 | 6.44 | 0.25 |
| Sufite | (NH4)2 SO3 | 7.2 | 6.8 | 7.5 | 7.8 | 0.3 |
| DL-Tartrate | (NH4)2 C4 H4 O6 | 4.37 | 9.63 | 6.83 | 6.84 | 0.01 |
| P-Toluate | NH4 C8 H7 O2 | 4.37 | 9.63 | 6.83 | 7.08 | 0.25 |
| Ammonium ion salts | Truncated estimated pH values | Truncated measured pH scale |
|---|---|---|
| Acetate | 7.0 | 6.9 |
| Benzoate | 6.8 | 6.6 |
| Citrate | 6.9 | 7.2 |
| Malonate | 7.1 | 7.1 |
| 1-Naphthyacetate | 6.8 | 7.0 |
| Oxolate | 6.6 | 6.4 |
| Sufite | 7.5 | 7.8 |
| Decilitre-Tartrate | 6.8 | 6.8 |
| P-Toluate | 6.8 | 7.1 |
Conclusion
The application of this simple technique is limited in application in a manner reminiscent to that of the Ideal Gas Police force. Nevertheless, this go up to pH scale calculations is an introduction for students to the quite interlinking nature of equilibrium between Brønsted-Lowry acids and bases and also allows for a discussion into an expanse not taught in secondary education and eldest year university chemical science.
Manus Monroe is a lecturer in chemistry at Sonoma State University, California, US.
calculate the ph of a 42 m nh4cl solution
Source: https://edu.rsc.org/maths/estimating-ph-of-ammonium-solutions-containing-weak-br%C3%B8nsted-lowry-bases/2020321.article
Posting Komentar untuk "calculate the ph of a 42 m nh4cl solution"