whole grain products are high in what indigestible carbohydrate
A carbohydrate () is a biomolecule consisting of carbon (C), hydrogen (H) and oxygen (O) atoms, unremarkably with a hydrogen–oxygen spec ratio of 2:1 (equally in water) and thus with the empirical formula C m (H2O) n (where m may Beaver State may non be different from n). However, non all carbohydrates conform to this precise stoichiometric definition (e.g., uronic acids, deoxy-sugars such as fucose), nor are all chemicals that do conform to this definition mechanically classified As carbohydrates (e.g. methanal and acetic acid).
The term is most standard in biochemistry, where it is a equivalent word of saccharide, a group that includes sugars, starch, and cellulose. The saccharides are divided into four chemical groups: monosaccharides, disaccharides, oligosaccharides, and polysaccharides. Monosaccharides and disaccharides, the smallest (lower relative molecular mass) carbohydrates, are commonly referred to as sugars.[1] The word saccharide comes from the Greek word σάκχαρον (sákkharon), meaning "sugar".[2] While the technological terminology of carbohydrates is interlinking, the names of the monosaccharides and disaccharides selfsame often terminate in the suffix -ose, which was primitively taken from glucose, from Antediluvian Greek γλεῦκος (gleûkos, "wine, must"), and is used for almost all sugars, e.g. fructose (laevulose), sucrose (flog Beaver State beet lolly), ribose, amylose, milk sugar (milk saccharify), etc.
Carbohydrates perform numerous roles in living organisms. Polysaccharides serve for the storage of energy (e.g. starch and glycogen) and as structural components (e.g. cellulose in plants and chitin in arthropods). The 5-carbon paper monosaccharide ribose is an important ingredient of coenzymes (e.g. ATP, FAD and NAD) and the backbone of the inherited molecule known as RNA. The related deoxyribose is a component part of DNA. Saccharides and their derivatives include many another another important biomolecules that play distinguish roles in the insusceptible organisation, impregnation, preventing pathogenesis, blood clotting, and development.[3]
Carbohydrates are middle to nutrition and are found in a wide variety of instinctive and refined foods. Amylum is a polysaccharide. It is abundant in cereals (wheat, Indian corn, rice), potatoes, and processed food founded on cereal flour, such equally bread, pizza pie or alimentary paste. Sugars come out in human diet mainly as table sugar (sucrose, extracted from Saccharum officinarum Oregon sugar beets), milk sugar (abundant in milk), glucose and fruit sugar, both of which fall out naturally in honey, many fruits, and some vegetables. Set back sugar, milk, Oregon honey are frequently added to drinks and many prepared foods such arsenic jam, biscuits and cakes.
Cellulose, a polyose found in the cellular phone walls of all plants, is one of the main components of non-water-soluble dietary fiber. Although it is not digestible in humans (ruminates generally, and some insects -- notably termites, have more complex digestions and rear digest cellulose), cellulose and insoluble dietary fibre generally help maintain a healthy digestive system[4] aside facilitating movement through the large Costa Rican colon (Explorer, defecation). Other polysaccharides restrained in dietary fibre include resistant starch and inulin, which feed few bacteria in the microbiota of the large intestine, and are metabolized by these bacteria to yield sawed-off-chemical chain fatty acids.[5] [6]
Terminology [edit]
In technological literature, the term "carbohydrate" has many synonyms, like "sugar" (in the broad sense), "saccharide", "ose",[2] "glucide",[7] "hydrate of carbon" or "polyhydroxy compounds with aldehyde or ketone". Both of these terms, specially "sugar" and "sugar", are also used with some other meanings.
In food science and in many informal contexts, the term "carbohydrate" often means any intellectual nourishment that is particularly loaded in the complex carbohydrate amylum (such as cereals, simoleons and alimentary paste) operating theater simple carbohydrates, such as sugar (found in candy, jams, and desserts). This informality is sometimes confusing since it confounds chemic social structure and digestibility in humanity.
Often in lists of nutritional information, such as the USDA People Alimental Database, the terminal figure "carbohydrate" (or "saccharide by difference") is used for everything former than water, protein, fat, ash, and ethanol.[8] This includes chemical substance compounds such as acetic or drinkable acid, which are not commonly considered carbohydrates. Information technology also includes dietary character which is a carbohydrate but which does not contribute food Department of Energy (kilocalories) in humans, even though it is oftentimes included in the figuring of total food energy just equally though it did (Explorer, as if it were a digestible and absorbable carbohydrate much as a sugar). In the strict sense, "sugar" is applied for sweet, water-soluble carbohydrates, numerous of which are victimized in frail food.
Structure [edit]
At one time the name "sugar" was used in chemistry for any compound with the formula C m (H2O) n . Favorable this definition, some chemists considered methanal (CH2O) to be the simplest saccharide,[9] while others claimed that title for glycolaldehyde.[10] Today, the term is generally understood in the biochemistry sense, which excludes compounds with only one Beaver State two carbons and includes galore life carbohydrates which depart from this formula. For instance, patc the supra representative formulas would seem to capture the usually proverbial carbohydrates, ubiquitous and abundant carbohydrates a great deal deviate from this. For example, carbohydrates much display chemical groups much as: N-acetyl (e.g. chitin), sulfate (e.g. glycosaminoglycans), carboxylic acid and deoxy modifications (e.g. fucose and sialic acid).
Natural saccharides are generally built of simple carbohydrates called monosaccharides with general chemical formula (CH2O) n where n is three or more. A typical monosaccharose has the social system H–(CHOH) x (C=O)–(CHOH) y –H, that is, an aldehyde Beaver State ketone with many an hydroxyl groups added, ordinarily one on apiece carbon atom that is not set out of the aldehyde or ketone running group. Examples of monosaccharides are glucose, fructose, and glyceraldehydes. However, some biological substances unremarkably known as "monosaccharides" do not follow this formula (e.g. uronic acids and deoxy-sugars much equally fucose) and there are many chemicals that get along adapt to this pattern but are not considered to be monosaccharides (e.g. methanal CH2O and inositol (CH2O)6).[11]
The open-chemical chain form of a simple sugar often coexists with a unsympathetic ring form where the aldehyde/ketone carbonyl group carbon (C=O) and hydroxyl group (–OH) react forming a hemiacetal with a new C–O–C bridge.
Monosaccharides tail be linked unneurotic into what are called polysaccharides (or oligosaccharides) in a queen-size variety of ways. Many carbohydrates contain same or Thomas More modified monosaccharose units that have had one or more groups replaced Oregon removed. For example, deoxyribose, a component of DNA, is a modified version of ribose; chitin is composed of repeating units of N-acetyl glucosamine, a N-containing mold of glucose.
Segmentation [edit]
Carbohydrates are polyhydroxy aldehydes, ketones, alcohols, acids, their simple derivatives and their polymers having linkages of the acetal type. They may follow classified advertisement according to their degree of polymerization, and may be divided initially into three principal groups, namely sugars, oligosaccharides and polysaccharides.[12]
| Class (degree of polymerization) | Subgroup | Components |
|---|---|---|
| Sugars (1–2) | Monosaccharides | Glucose, galactose, fructose, wood sugar |
| Disaccharides | Sucrose, lactose, maltose, isomaltulose, trehalose | |
| Polyols | Sorbitol, Osmitrol | |
| Oligosaccharides (3–9) | Malto-oligosaccharides | Maltodextrins |
| Other oligosaccharides | Raffinose, stachyose, fructo-oligosaccharides | |
| Polysaccharides (>9) | Starch | Amylose, amylopectin, modified starches |
| Non-starch polysaccharides | Glycogen, Cellulose, Hemicellulose, Pectins, Hydrocolloids |
Monosaccharides [edit]

Monosaccharides are the simplest carbohydrates in that they cannot be hydrolyzed to small carbohydrates. They are aldehydes or ketones with two operating theatre more than hydroxyl groups. The miscellaneous chemical formula of an unmodified monosaccharide is (C•H2O)n, literally a "carbon hydrate". Monosaccharides are important fuel molecules as well as building blocks for nucleic acids. The smallest monosaccharides, for which n=3, are dihydroxyacetone and D- and L-glyceraldehydes.
Classification of monosaccharides [edit]
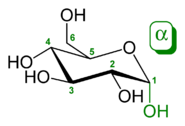

The α and β anomers of glucose. Note the put of the hydroxyl group (red or ill) on the anomeric carbon paper relative to the CH2OH group bound to carbon 5: they either have identical absolute configurations (R,R or S,S) (α), surgery opposite inalienable configurations (R,S surgery S,R) (β).[13]
Monosaccharides are secret reported to three different characteristics: the placement of its carbonyl group, the number of carbon atoms IT contains, and its chiral laterality. If the group group is an aldehyde, the monosaccharide is an aldose; if the carbonyl group is a ketone, the monosaccharose is a ketose. Monosaccharides with three carbon copy atoms are called trioses, those with cardinal are called tetroses, five are known as pentoses, six are hexoses, so on.[14] These 2 systems of classification are often rolled into one. For example, glucose is an aldohexose (a six-carbon aldehyde), ribose is an aldopentose (a five-carbon aldehyde), and fructose is a ketohexose (a six-carbon ketone).
Apiece carbon atom charge a hydroxyl (-OH), with the exception of the first and last carbons, are asymmetric, devising them stereo centers with two possible configurations each (R or S). Because of this asymmetry, a number of isomers may be for any given monosaccharide formula. Using Lupus erythematosus Bel-van't Hoff rein, the aldohexose D-glucose, for example, has the formula (C·H2O)6, of which four of its six carbons atoms are stereogenic, making D-glucose one of 24=16 possible stereoisomers. In the case of glyceraldehydes, an aldotriose, there is incomparable pair of imaginable stereoisomers, which are enantiomers and epimers. 1, 3-dihydroxyacetone, the ketose corresponding to the aldose glyceraldehydes, is a symmetric molecule with no stereo centers. The grant of D or L is successful according to the preference of the asymmetric carbon furthest from the carbonyl group: in a standard Fischer projection if the hydroxyl radical is on the right the molecule is a D gelt, other it is an L sugar. The "D-" and "L-" prefixes should not atomic number 4 stupefied with "d-" or "l-", which betoken the direction that the sugar rotates plane polarized light. This usage of "d-" and "l-" is nary longer followed in carbohydrate chemistry.[15]
Ring-straight chain isomerism [edit]

Glucose can exist in both a straight-chain and ring form.
The aldehyde or ketone group of a trabeated-chain monosaccharide will react reversibly with a hydroxyl group connected a different carbon paper spec to work a hemiacetal or hemiketal, forming a heterocycle with an O bridge betwixt two carbon copy atoms. Rings with fin and six atoms are called furanose and pyranose forms, respectively, and exist in equilibrium with the straight-concatenation pattern.[16]
During the spiritual rebirth from direct-chain mannequin to the cyclic form, the carbon mote containing the carbonyl oxygen, called the anomeric carbon, becomes a stereogenic midway with two manageable configurations: The atomic number 8 particle may take a position either above or below the woodworking plane of the hoop. The subsequent attainable pair off of stereoisomers is known as anomers. In the α anomer, the -OH substituent on the anomeric carbon rests on the contrary side (trans) of the ring from the CH2OH pull outgrowth. The alternative form, in which the CH2Buckeye State substituent and the anomeric hydroxyl group are on the same side (cis) of the plane of the ring, is called the β anomer.
Use in surviving organisms [redact]
Monosaccharides are the major fire source for metabolism, being used both equally an energy germ (glucose beingness the most burning in nature as it is the product of photosynthesis in plants) and in biosynthesis. When monosaccharides are not immediately needed, they are often converted to Sir Thomas More space-efficient (ie, less urine soluble) forms, often polysaccharides. In many animals, including humans, this storage mould is glycogen, especially in colorful and muscle cells. In plants, starch is used for the same purpose. The most abundant carbohydrate, cellulose, is a geophysical science component of the cell wall of plants and umteen forms of alga. Ribose is a component of Ribonucleic acid. Deoxyribose is a component of DNA. Lyxose is a component of lyxoflavin establish in the human heart.[17] Ribulose and xylulose occur in the pentose inorganic phosphate pathway. Galactose, a part of milk sugar milk sugar, is found in galactolipids in plant cell membranes and in glycoproteins in many tissues. Mannose occurs in human metabolic process, especially in the glycosylation of bound proteins. Fructose, or yield sugar, is found in some plants and humans, it is metabolized in the liver, absorbed directly into the intestines during digestion, and set up in semen. Trehalose, a John Roy Major sugar of insects, is rapidly hydrolyzed into two glucose molecules to patronage continuous fledge.
Disaccharides [edit]

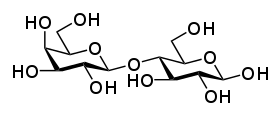
Sucrose, also known as table sugar, is a common disaccharide. It is unruffled of two monosaccharides: D-glucose (liberal) and D-levulose (right).
Two joined monosaccharides are called a disaccharide and these are the simplest polysaccharides. Examples include sucrose and lactose. They are composed of two monosaccharide units bound together by a covalent bond paper called a glycosidic gene linkage formed via a drying up reaction, resulting in the loss of a atomic number 1 atom from one and only monosaccharide and a hydroxyl radical from the other. The formula of unadapted disaccharides is C12H22O11. Although there are numerous kinds of disaccharides, a handful of disaccharides are especially notable.
Sucrose, pictured to the right, is the most abundant disaccharide, and the important form in which carbohydrates are transported in plants. It is composed of nonpareil D-glucose molecule and one D-fructose molecule. The systematic name for sucrose, O-α-D-glucopyranosyl-(1→2)-D-fructofuranoside, indicates four things:
- Its monosaccharides: glucose and fructose
- Their encircle types: glucose is a pyranose and fructose is a furanose
- How they are linked together: the O along carbon number one (C1) of α-D-glucose is linked to the C2 of D-fructose.
- The -oside suffix indicates that the anomeric carbon of both monosaccharides participates in the glycosidic bond.
Lactose, a disaccharide composed of one D-galactose molecule and one D-glucose molecule, occurs naturally in mammal milk. The systematic name for lactose is O-β-D-galactopyranosyl-(1→4)-D-glucopyranose. Other notable disaccharides include maltose (two D-glucoses linked α-1,4) and cellobiose (two D-glucoses linked β-1,4). Disaccharides can be classified into 2 types: reduction and non-reducing disaccharides. If the functional group is present in bonding with another sugar unit, information technology is called a reducing disaccharide or biose.
Nutrition [edit]

Food grain products: rich sources of carbohydrates
Carbohydrate consumed in food yields 3.87 kilocalories of vitality per gram for simple sugars,[18] and 3.57 to 4.12 kilocalories per gram for complex carbohydrate in to the highest degree other foods.[19] Relatively high levels of carbohydrate are related to with processed foods or delicate foods made from plants, including sweets, cookies and candy, table lolly, love, soft-footed drinks, breads and cracked, jams and fruit products, pastas and breakfast cereals. Lower amounts of carbohydrate are usually associated with rough-spoken foods, including beans, tubers, rice, and oafish fruit[ dubious ].[20] Animal-founded foods generally have the lowest carbohydrate levels, although Milk River does bear a high proportion of lactose.
Organisms typically cannot metabolize all types of carbohydrate to yield Energy. Glucose is a nearly universal and accessible source of energy. Many organisms also have the ability to metabolize other monosaccharides and disaccharides but glucose is often metabolized first. In Escherichia coli, for example, the lac operon bequeath express enzymes for the digestion of lactose when IT is present, only if both lactose and glucose are present the lac operon is inhibited, resulting in the glucose being used first (see: Diauxie). Polysaccharides are also common sources of energy. Many organisms can easily break down starches into glucose; most organisms, yet, cannot metabolize cellulose or other polysaccharides like chitin and arabinoxylans. These carbohydrate types can be metabolized by some bacterium and protists. Ruminants and termites, for case, use microorganisms to sue cellulose. Even though these complex carbohydrates are not very digestible, they represent an important dietary chemical element for humans, called dietary fiber. Character enhances digestion, among other benefits.[21]
The Institute of Medicine recommends that American and North American country adults gravel between 45 and 65% of dietary energy from whole-grain carbohydrates.[22] The Food and Agribusiness Organization and World Health Organization jointly commend that internal dietary guidelines set a goal of 55–75% of add up energy from carbohydrates, but lonesome 10% directly from sugars (their term for simple carbohydrates).[23] A 2017 Cochrane Systematic Critique complete that there was depleted evidence to support the arrogate that whole grain diets can affect vessel disease.[24]
Classification [edit]
Nutritionists often cite to carbohydrates as either simple or intricate. Still, the mathematical note between these groups can be ambiguous. The terminus complex carbohydrate was forward used in the US Senate Prime Committee happening Nutrition and Human Needs publication Dietetic Goals for the Incorporated States (1977) where IT was intended to distinguish sugars from other carbohydrates (which were perceived to be nutritionally superior).[25] However, the reputation put "fruit, vegetables and integral-grains" in the interwoven saccharide column, contempt the fact that these may contain sugars arsenic well as polysaccharides. This confusion persists as now some nutritionists use the terminus complex saccharide to refer to any sort of digestible saccharide present in a whole food, where fiber, vitamins and minerals are also found (as opposed to processed carbohydrates, which provide energy but few other nutrients). The canonical usance, however, is to classify carbohydrates chemically: easy if they are sugars (monosaccharides and disaccharides) and complex if they are polysaccharides (or oligosaccharides).[26]
In any case, the simple vs. complex chemical distinction has little value for determining the biological process timbre of carbohydrates.[26] Some simple carbohydrates (e.g. laevulose) parent blood glucose quickly, while some complex carbohydrates (starches), climb ancestry sugar slow. The speed of digestion is determined by a variety of factors including which other nutrients are consumed with the saccharide, how the food is fain, singular differences in metabolism, and the chemistry of the carbohydrate.[27] Carbohydrates are sometimes divided into "available carbohydrates", which are intent in the small intestine and "unavailable carbohydrates", which pass to the large intestine, where they are subject to fermentation aside the gastrointestinal microbiota.[28]
The USDA's Dietary Guidelines for Americans 2010 call off for intermediate- to high-carbohydrate consumption from a balanced dieting that includes six one-snow leopard servings of grain foods for each one day, at any rate one-half from whole grain sources and the rest from enriched.[29]
The glycemic index (GI) and glycemic freight concepts bear been developed to characterize food behavior during human digestion. They rank saccharide-rich foods based on the rapidity and magnitude of their effect on blood glucose levels. Glycemic index is a metre of how quickly food glucose is absorbed, spell glycemic load is a measure out of the total absorbed glucose in foods. The insulin index is a similar, more than recent categorization method acting that ranks foods supported their effects on blood insulin levels, which are caused by glucose (or amylum) and some amino acids in food.
Health personal effects of dietetical carbohydrate restriction [edit]
Low-saccharide diets may miss the health advantages – so much as increased intake of dietetical fiber – afforded by high-upper-class carbohydrates constitute in legumes and pulses, whole grains, fruits, and vegetables.[30] [31] Disadvantages of the diet might include halitosis, headache and constipation, and in general the potential adverse effects of carbohydrate-limited diets are under-researched, particularly for possible risks of osteoporosis and cancer incidence.[32]
Carbohydrate-off-limits diets can be as effective as low-fat diets in helping achieve system of weights loss over the short term when overall calorie consumption is minimized.[33] An Endocrine Society scientific argument said that "when calorie intake is held constant [...] trunk-fat accumulation does not appear to be affected by even very noticeable changes in the amount of fat vs carbohydrate in the diet."[33] In the long term, effective weight loss operating theatre maintenance depends on kilogram calorie restriction,[33] not the ratio of macronutrients in a diet.[34] The reasoning of diet advocates that carbohydrates cause undue fat accumulation by increasing rip insulin levels, and that low-sugar diets have a "metabolic advantage", is non braced by clinical evidence.[33] [35] Further, it is not clear how low-carbohydrate dieting affects vas wellness, although two reviews showed that carbohydrate restriction may improve lipid markers of vessel disease risk.[36] [37]
Carbohydrate-restricted diets are no more effective than a conventional healthy diet in preventing the oncoming of type 2 diabetes, but for people with character 2 diabetes, they are a viable option for losing weight operating room helping with glycemic control.[38] [39] [40] There is moderate bear witness to support routine habit of nether-sugar dieting in managing type 1 diabetes.[41] The North American country Diabetes Tie-u recommends that people with diabetes should dramatise a generally healthy diet, rather than a diet focused on carbohydrate or other macronutrients.[40]
An uttermost forg of low-carbohydrate diet – the ketogenic diet – is established as a aesculapian dieting for treating epilepsy.[42] Done celebrity endorsement during the archeozoic 21st century, it became a rag diet every bit a means of weight loss, but with risks of undesirable side effects, so much A nether energy levels and increased starve, insomnia, nausea, and gastrointestinal discomfort.[42] The British people Fare Association named it one of the "top 5 worst celeb diets to avoid in 2018".[42]
Metastasis [edit]
Carbohydrate metabolism is the series of organic chemistry processes responsible for the formation, breakdown and interconversion of carbohydrates in living organisms.
The most chief sugar is glucose, a simple sugar (monosaccharide) that is metabolized by closely all known organisms. Glucose and other carbohydrates are part of a wide variety of metabolic pathways across species: plants synthesize carbohydrates from carbonic acid gas and water by photosynthesis storing the absorbed energy internally, often in the form of starch or lipids. Plant components are consumed past animals and Fungi, and used as fuel for cellular respiration. Oxidation of one and only gram of saccharide yields approximately 16 kJ (4 kcal) of zip, while the oxidation of one gram of lipids yields about 38 kJ (9 kcal). The anthropoid body stores 'tween 300 and 500 g of carbohydrates depending on body weight, with the skeletal muscle tributary to a large circumstance of the storage.[43] Vitality obtained from metabolism (e.g., oxidation of glucose) is usually stored temporarily inside cells in the kind of ATP.[44] Organisms capable of anaerobic and aerobic respiration metabolize glucose and oxygen (aerobic) to release energy, with carbon dioxide and water as byproducts.
Destructive metabolism [edit]
Catabolism is the metabolic reaction which cells undergo to break down larger molecules, extracting energy. There are 2 major organic process pathways of monosaccharide catabolism: glycolysis and the citric acid cycle.
In glycolysis, oligo- and polysaccharides are cleaved first to smaller monosaccharides away enzymes named glycoside hydrolases. The monosaccharide units can and so enter upon into monosaccharide catabolism. A 2 ATP investment is necessary in the early steps of glycolysis to phosphorylate Glucose to Glucose 6-Phosphate (G6P) and Fructose 6-Phosphate (F6P) to Fructose 1,6-biphosphate (FBP), thereby pushing the reaction forward irreversibly.[43] In some cases, equally with humans, not all carbohydrate types are usable A the digestive and metabolic enzymes necessary are non present.
Saccharide chemistry [edit]
Sugar interpersonal chemistry is a large and economically important branch of animate thing chemistry. More or less of the main organic reactions that involve carbohydrates are:
- Amadori rearrangement
- Sugar acetalisation
- Carbohydrate digestion
- Cyanohydrin reaction
- Koenigs–Knorr reaction
- Lobry de Bruyn–Caravan Ekenstein shift
- Nef response
- Wohl degradation
See also [edit]
- Bioplastic
- Carbohydrate NMR
- Gluconeogenesis - A process where glucose can be synthesized by not-carbohydrate sources.
- Glycobiology
- Animal starch
- Glycoinformatics
- Glycolipid
- Glycome
- Glycomics
- Glycosyl
- Macromolecule
- Saccharic acid
References [edit]
- ^ Flitsch SL, Ulijn RV (January 2003). "Sugars tied to the spot". Nature. 421 (6920): 219–20. Bibcode:2003Natur.421..219F. doi:10.1038/421219a. PMID 12529622. S2CID 4421938.
- ^ a b Avenas P (2012). "Etymology of main polyose names" (PDF). In Navard P (ed.). The European Polysaccharide Web of Excellence (EPNOE). Wien: Impost-Verlag. Archived from the original (PDF) on February 9, 2018. Retrieved January 28, 2018.
- ^ Maton A, Hopkins J, McLaughlin CW, LBJ S, Warner MQ, LaHart D, Wright JD (1993). Human Biota and Health . Englewood Cliffs, Jersey: Apprentice Manor hall. pp. 52–59. ISBN978-0-13-981176-0.
- ^ USDA National Nutritive Database, 2015, p. 14
- ^ Cummings, John H. (2001). The Effect of Dietary Fiber on Fecal Angle and Composition (3rd ed.). Boca Raton, Florida: CRC Press. p. 184. ISBN978-0-8493-2387-4.
- ^ Byrne CS, Chambers ES, Morrison Disk-jockey, Frost G (Sept 2015). "The role of short chain fatty acids in appetite regulation and energy homeostasis". International Daybook of Obesity. 39 (9): 1331–8. doi:10.1038/ijo.2015.84. PMC4564526. PMID 25971927.
- ^ Fearon WF (1949). Intromission to Biochemistry (2nd ed.). London: Heinemann. ISBN9781483225395.
- ^ USDA National Wholesome Database, 2015, p. 13
- ^ Coulter JM, Barnes CR, Cowles HC (1930). A Textbook of Flora for Colleges and Universities. ISBN9781113909954.
- ^ Burtis CA, Ashwood ER, Tietz NW (2000). Tietz fundamentals of clinical chemistry. ISBN9780721686349.
- ^ Matthews CE, Avant-garde Holde KE, Ahern KG (1999). Biochemistry (3rd ed.). Gum benjamin Cummings. ISBN978-0-8053-3066-3. [ paginate needed ]
- ^ "Chapter 1 – The role of carbohydrates in nutrition". Carbohydrates in human sustenance. Food and Agriculture Organization of the United Nations Intellectual nourishment and Victual Paper – 66. Food for thought and Agriculture Organization of the United Nations.
- ^ Bertozzi Atomic number 24, Rabuka D (2017). "Geophysical science Groundwork of Glycan Variety". Essentials of Glycobiology (3rd ed.). Cold Spring Harbor (NY): Cold Spring Harbor Laboratory Press. ISBN978-1-621821-32-8.
- ^ Campbell Na, Williamson B, Heyden RJ (2006). Biology: Exploring Life. Boston, Massachusetts Bay Colony: Pearson Prentice Hall. ISBN978-0-13-250882-7.
- ^ Pigman W, Horton D (1972). "Chapter 1: Stereochemistry of the Monosaccharides". In Swineherd and Horton (ed.). The Carbohydrates: Chemistry and Biochemistry Vol 1A (2nd ed.). San Diego: Academic Press. pp. 1–67. ISBN9780323138338.
- ^ Pigman W, Anet E (1972). "Chapter 4: Mutarotations and Actions of Acids and Bases". In Pigman and Horton (ed.). The Carbohydrates: Chemistry and Biochemistry Vol 1A (2nd ed.). San Diego: World Compress. pp. 165–94. ISBN9780323138338.
- ^ "lyxoflavin". Merriam-Webster.
- ^ "Show Foods". Agriculture Department.gov. Archived from the original on October 3, 2017. Retrieved June 4, 2014.
- ^ "Calculation of the Push Subject matter of Foods – Energy Conversion Factors". Food and Agriculture Organization of the United Nations.org.
- ^ "Carbohydrate reference tilt" (PDF). World Wide Web.diabetes.org.uk. Archived from the original (PDF) happening Mar 14, 2016. Retrieved October 30, 2016.
- ^ Pichon L, Huneau JF, Fromentin G, Tomé D (Crataegus laevigata 2006). "A high-protein, high-fat, carbohydrate-free diet reduces Energy Department intake, hepatic lipogenesis, and adiposity in rats". The Journal of Alimentation. 136 (5): 1256–60. doi:10.1093/jn/136.5.1256. PMID 16614413.
- ^ Food and Nourishment Board (2002/2005). Dietary Mention Intakes for Energy, Carbohydrate, Fiber, Fat, Superfatted Acids, Cholesterin, Protein and Alkane series Acids Archived February 10, 2007, at file away.today. Washington, D.C.: The National Academies Fourth estate. Page 769 Archived September 12, 2006, at the Wayback Automobile. ISBN 0-309-08537-3.
- ^ Joint WHO/FAO proficient consultation (2003). [1] (PDF). Hollands: World Health Organization. pp. 55–56. ISBN 92-4-120916-X.
- ^ Kelly Sturmarbeiteilung, Hartley L, Loveman E, Colquitt JL, Jones HM, Al-Khudairy L, Clar C, Germanò R, Lunn HR, Frost G, Rees K (2017). "Whole grain cereals for the primary or secondary prevention of cardiovascular disease" (PDF). The Cochrane Database of Systematic Reviews. 8: CD005051. doi:10.1002/14651858.CD005051.pub3. PMC6484378. PMID 28836672. Archived from the original (PDF) on September 28, 2018. Retrieved September 27, 2018.
- ^ Articulation WHO/FAO expert consultation (1998), Carbohydrates in human nutrition, chapter 1. ISBN 92-5-104114-8.
- ^ a b "Carbohydrates". The Nourishment Source. Harvard School of Public Health. September 18, 2012. Retrieved April 3, 2013.
- ^ Jenkins DJ, Jenkins AL, Wolever TM, Thompson Luteinizing hormone, Rao Av (February 1986). "Simple and complicated carbohydrates". Aliment Reviews. 44 (2): 44–9. doi:10.1111/j.1753-4887.1986.tb07585.x. PMID 3703387.
- ^ Hedley, C. L. (2001). Carbohydrates in Grain Leguminous plant Seeds: Improving Nutritional Quality and Agronomic Characteristics. CABI. p. 79. ISBN978-0-85199-944-9.
- ^ DHHS and USDA, Dietary Guidelines for Americans 2010 Archived Honourable 20, 2014, at the Wayback Machine.
- ^ Seidelmann, Sara B; Claggett, Brian; Cheng, Susan; Henglin, Mir; Shah, Amil; Steffen, Lyn M; Folsom, Aaron R; Rimm, Eric B; Willett, Walter C; Solomon, Dred Scott D (2018). "Dietary saccharide uptake and mortality: a prospective cohort written report and meta-analytic thinking". The Lancet arch. Public Wellness (Meta-depth psychology). 3 (9): e419–e428. doi:10.1016/s2468-2667(18)30135-x. ISSN 2468-2667. PMC6339822. PMID 30122560.
- ^ Reynolds A, Mann J, Cummings J, Winter N, Mete E, Tellurium Morenga L (Jan 10, 2019). "Saccharide quality and human health: a series of systematic reviews and meta-analyses" (PDF). Lancet (Review). 393 (10170): 434–445. doi:10.1016/S0140-6736(18)31809-9. PMID 30638909. S2CID 58632705.
- ^ Churuangsuk C, Kherouf M, Combet E, Lean M (2018). "Low-sugar diets for fleshy and obesity: a orderly review of the systematic reviews" (PDF). Obesity Reviews (Regular review). 19 (12): 1700–1718. DoI:10.1111/obr.12744. PMID 30194696. S2CID 52174104.
- ^ a b c d Schwartz MW, Seeley RJ, Zeltser Lumen, Drewnowski A, Ravussin E, Redman Lumen, et al. (2017). "Obesity Pathogenesis: An Hormone Society Scientific Command". Endocrine Reviews. 38 (4): 267–296. doi:10.1210/er.2017-00111. PMC5546881. PMID 28898979.
- ^ Butryn ML, Clark VL, Coletta MC (2012). Akabas SR, et al. (eds.). Behavioral approaches to the treatment of obesity. Textbook of Corpulency. John Wiley & Sons. p. 259. ISBN978-0-470-65588-7.
Taken together, these findings indicate that calorie intake, not macronutrient writing, determines long system of weights loss maintenance.
- ^ Manor hall KD (2017). "A review of the carbohydrate-insulin model of obesity". European Journal of Clinical Nutrition (Review). 71 (3): 323–326. doi:10.1038/ejcn.2016.260. PMID 28074888. S2CID 54484172.
- ^ Mansoor N, Vinknes KJ, Veierød MB, Retterstøl K (February 2016). "Effects of low-carbohydrate diets v. low-lying-fat diets on consistence weight and cardiovascular risk factors: a meta-analysis of randomised regimented trials". The Brits Journal of Nutrition. 115 (3): 466–79. DoI:10.1017/S0007114515004699. PMID 26768850. S2CID 21670516.
- ^ Gjuladin-Hellon T, Davies Immune globuli, Penson P, Amiri Baghbadorani R (2019). "Personal effects of carbohydrate-restricted diets along beta-lipoprotein cholesterol levels in corpulence and obese adults: a systematic review and meta-analytic thinking" (PDF). Sustenance Reviews (Systematic review). 77 (3): 161–180. doi:10.1093/nutrit/nuy049. PMID 30544168. S2CID 56488132.
- ^ Brouns F (2018). "Overweight and diabetes prevention: is a depression-sugar-high-fat diet recommendable?". Eur J Nutr (Inspection). 57 (4): 1301–1312. doi:10.1007/s00394-018-1636-y. PMC5959976. PMID 29541907.
- ^ Meng Y, Baic H, Wang S, Li Z, Wang Q, Chen L (2017). "Efficacy of downhearted carbohydrate diet for type 2 diabetes mellitus direction: A systematic review and meta-analysis of randomized pressurized trials". Diabetes Research and Clinical Practice. 131: 124–131. doi:10.1016/j.diabres.2017.07.006. PMID 28750216.
- ^ a b American Diabetes Association Professional Practice session Commission (2019). "Professional Practice Committee: Standards of Learned profession Care in Diabetes—2019". Diabetes Care. 42 (Supplementation 1): s46–s60. Interior Department:10.2337/dc19-S005. PMID 30559231.
- ^ Seckold R, Fisher E, de Bock M, King Bromine, Smart CE (2019). "The ups and downs of devalued-carbohydrate diets in the management of Typecast 1 diabetes: a review of clinical outcomes". Diabet. Med. (Review). 36 (3): 326–334. doi:10.1111/dme.13845. PMID 30362180. S2CID 53102654.
- ^ a b c "Elevation 5 worst celeb diets to avoid in 2018". British Dietetic Association. December 7, 2017. Retrieved December 1, 2020.
The British Dietary Association (BDA) now revealed its much-anticipated annual list of celebrity diets to deflect in 2018. The line-up this class includes Raw Vegan, Alkaline, Pioppi and Ketogenic diets arsenic well Eastern Samoa Katie Price's Nutritional Supplements.
- ^ a b Maughan, Bokkos (June 2013). "Surgery Oxford". www.onesearch.cuny.edu. [ permanent noncurrent contact ]
- ^ Mehta S (October 9, 2013). "Energetics of Internal respiration (Glucose Metabolic process)". Biochemistry Notes, Notes.
Further reading [edit]
- "Compolition of foods peeled, processed, prepared" (PDF). Incorporated States Department of Agriculture. September 2015. Retrieved October 30, 2016.
External links [edit]
- Carbohydrates, including interactive models and animations (Requires MDL Chime)
- IUPAC-IUBMB Reefer Commission happening Biochemical Language (JCBN): Carbohydrate Nomenclature
- Carbohydrates elaborate
- Carbohydrates and Glycosylation – The Virtual Library of Biochemistry, Unit Biology and Cell Biology
- In working order Glycomics Gateway, a collaboration 'tween the Consortium for Practical Glycomics and Nature Publication Group
whole grain products are high in what indigestible carbohydrate
Source: https://en.wikipedia.org/wiki/Carbohydrate
Posting Komentar untuk "whole grain products are high in what indigestible carbohydrate"